What was the challenge or problem to be solved?
Ozone disinfection in industrial textiles
The project originated from the need to ensure reliable microbial reduction across different textile substrates used in demanding industrial environments. The manufacturer aimed to introduce a solution capable of delivering consistent hygienic performance while maintaining the structural and functional integrity of treated materials.
Although ozone is widely recognized for its oxidizing and antimicrobial properties, its real effectiveness depends on exposure time, concentration levels, gas distribution and the interaction with the specific textile matrix. Variations in fiber composition, porosity and humidity can significantly influence treatment efficiency.
Ozone performance varies depending on textile type and exposure conditions.
For this reason, experimental verification was required to confirm measurable performance rather than relying on theoretical assumptions. The objective was to determine whether the ozone disinfection process could achieve significant microbial load reduction under realistic operating scenarios.
The expected outcome was a technically supported solution aligned with industrial hygiene standards, capable of delivering repeatable and verifiable results.
Microbiological validation in ozone disinfection
The core objective involved rigorous microbiological validation to quantify antimicrobial performance using standardized assessment criteria. Qualitative observations were insufficient; the project required measurable and reproducible microbial load reduction supported by laboratory evidence.
Testing protocols were specifically designed to simulate real-use conditions, including representative contamination levels and exposure scenarios. This ensured that the evaluation of ozone disinfection reflected practical application rather than idealized laboratory conditions.
Controlled assays allowed comparison between treated and untreated samples, enabling precise calculation of microbial reduction levels. This data-driven approach minimized uncertainty and provided objective benchmarks for performance assessment.
Ultimately, the purpose was to define operational parameters capable of ensuring consistent antimicrobial efficacy across different material types and environmental contexts.
Technical evaluation of the disinfection device
The main technical difficulty involved understanding how operational variables influenced overall system performance. Gas flow dynamics, internal chamber configuration and environmental conditions such as humidity and temperature could significantly affect ozone distribution and contact efficiency.
INFINITIA conducted a comprehensive technical evaluation without modifying the core architecture of the device. The goal was to identify optimization opportunities while preserving the original engineering concept and ensuring operational safety.
Particular attention was given to potential inconsistencies in treatment homogeneity, especially in multilayer textile structures where ozone penetration may vary. The evaluation required a systematic assessment of exposure patterns and potential performance deviations.
Given the innovative nature of the device, limited benchmark data were available for comparison. This required a structured methodology combining controlled experimentation with applied engineering analysis to extract meaningful conclusions.
How was it addressed or what was the solution?
The intervention combined controlled microbiological testing with detailed operational analysis. The objective was to validate ozone disinfection performance under realistic conditions and define improvements supported by experimental data.
Optimization of ozone disinfection devices
An experimental plan was structured to identify critical performance factors influencing antimicrobial efficacy. Different ozone concentration ranges, exposure times and operational sequences were evaluated within safe and technically feasible limits.
The analysis revealed that relatively minor adjustments in exposure duration and concentration stability could significantly enhance microbial reduction outcomes. Rather than redesigning the equipment, optimization focused on refining the balance between ozone generation and effective contact time.
Small adjustments in concentration and exposure time can significantly improve effectiveness.
Environmental factors were also assessed to determine their impact on system efficiency. Variations in ambient humidity and textile load configuration were considered to better understand performance variability in real operating conditions.
This structured optimization approach strengthened the consistency of the ozone disinfection process and reduced potential performance fluctuations between cycles.
Validation of disinfection systems through controlled testing
The execution included tests specifically designed to measure microbial reduction on treated textiles and surfaces. To this end, representative standard materials were selected as experimental references, ensuring homogeneous and comparable conditions across trials.
The samples were prepared and inoculated with representative microorganisms such as E. coli and S. aureus in order to simulate real contamination scenarios. This approach made it possible to reproduce realistic usage conditions and ensure the technical relevance of the results obtained.
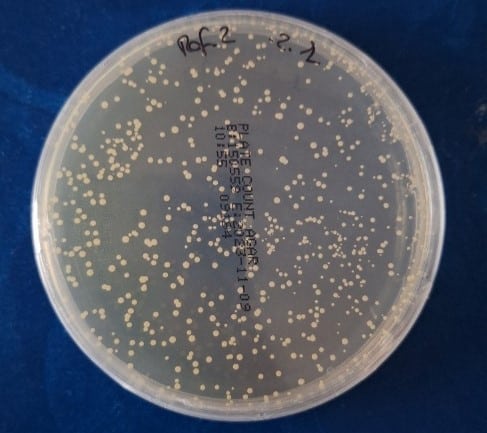
Following inoculation, different ozone treatment programs were applied, and the surviving bacteria were quantified using culture methods and microbiological analysis techniques. Colony-forming unit counts enabled the establishment of objective metrics to assess the effectiveness of each tested configuration.
Colony counting enables objective quantification of treatment effectiveness.
The comparative analysis between treated and untreated samples made it possible to determine the efficacy associated with each ozone concentration evaluated. This validation of disinfection systems was not limited to confirming the oxidizing action of the gas, but also provided insight into the overall device response under different operational scenarios.
The interpretation of the results was carried out from an applied technical perspective, transforming experimental data into clear recommendations for the manufacturer. In this way, the knowledge generated was directly integrated into the optimization and further development of the product.
Experimentally validated microbial load reduction
The study confirmed significant microbial load reduction under optimized operational conditions. Results demonstrated that effectiveness was not solely dependent on ozone generation capacity but on precise control of exposure dynamics and distribution uniformity.
Operational ranges were defined to maximize antimicrobial efficacy without compromising textile integrity or long-term material stability. This ensured that performance improvements did not introduce secondary risks.
Consistency testing across multiple cycles reinforced the reliability of the treatment process. The system showed stable behavior within defined parameters, reducing variability and increasing operational predictability.
Overall, the project transformed an early-stage technological concept into a validated industrial solution supported by experimental evidence, engineering analysis and quantified antimicrobial performance.