What was the challenge or problem to be solved?
In certain industrial sectors, the incorporation of materials with antimicrobial properties cannot rely solely on technical claims or preliminary testing. When these materials must operate in highly demanding applications, it becomes essential to validate their real performance under representative conditions of use.
In this context, the project focused on ensuring that the antimicrobial functionality remained stable over time and after the material had been subjected to different mechanical and environmental stresses.
Material homologation in critical applications
The starting point was the need to carry out material homologation for use in environments where microbiological load, frequent handling, and environmental conditions could compromise performance. In such applications, small changes in surface condition or composition may significantly affect antimicrobial efficacy.
It was not sufficient to verify the presence of an additive or surface treatment; it was necessary to demonstrate that the entire system maintained its functionality after repeated cycles of use, cleaning, or environmental exposure. The client required technical guarantees before validating the material for final product integration, as insufficient validation could lead to loss of performance, claims, or health-related risks.
The homologation process had to be supported by quantifiable and comparable data aligned with objective technical criteria. The expected benefit was to enable an informed decision that would reduce uncertainty in later industrialization stages.
Antimicrobial activity under demanding conditions
One of the key aspects was the evaluation of antimicrobial activity after subjecting the material to different controlled stress scenarios. In many cases, antimicrobial materials show favorable results under initial laboratory conditions, but their performance may be affected by surface wear, additive migration, or chemical degradation.
The project aimed to determine whether antimicrobial efficacy remained within acceptable ranges after simulated aging. To this end, conditions were defined to represent real accumulated use, including environmental exposure and repeated cycles.
Initial antimicrobial efficacy does not guarantee stability under real use: validating durability is key to preventing in-service failures.
The objective was not only to confirm a specific microbiological reduction at a given moment, but to analyze the stability of that reduction over time. This approach allowed validation to be addressed from a functional perspective rather than a purely declarative one, providing greater technical robustness to the process.
Accelerated testing as a technical challenge
The main challenge was to design an accelerated test capable of reproducing, within a reduced timeframe, the cumulative effects of prolonged use. This phase required defining parameters consistent with the final application, avoiding both underestimation and overrepresentation of real conditions.
The technical difficulty lay in establishing aging cycles that could anticipate potential degradation without artificially altering the material’s behavior. The representativeness of the applied conditions was critical to ensure that the results were extrapolable.
Poorly defined accelerated testing can lead to non-extrapolable results and incorrect technical decisions.
The INFINITIA team defined the stress scenarios and acceptance criteria, ensuring that the methodology combined scientific rigor with industrial applicability. This approach transformed initial uncertainty into a structured and objective evaluation process.
How was the challenge approached or what was the solution?
The solution was structured through a progressive methodology integrating use simulation, microbiological verification, and comparative analysis of results. The technical approach was oriented toward risk reduction prior to industrial implementation and toward ensuring consistency between testing conditions and the final application environment.
Custom testing for functional validation
Custom testing was designed specifically for the material’s application context. This customization made it possible to adjust critical variables such as temperature, relative humidity, handling cycles, or exposure to external agents, avoiding the use of standard protocols that did not reflect operational reality.
The approach included initial material characterization followed by evaluation after the defined stress conditions. This comparison made it possible to identify potential variations in antimicrobial performance and assess their technical relevance.
The functional validation strategy was not limited to a single scenario but incorporated different representative stress conditions. In this way, material validation was supported by a robust experimental framework oriented toward informed decision-making.
Microbiological testing and performance evaluation
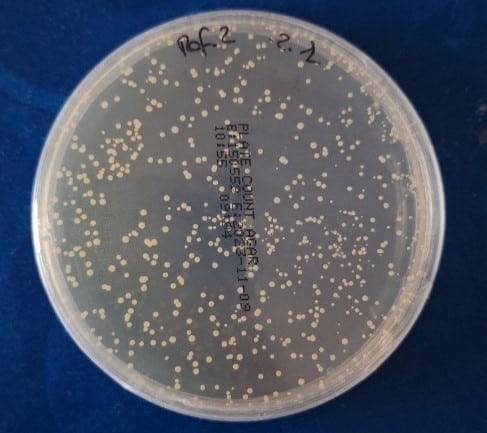
Efficacy verification was carried out through microbiological testing that quantified microbial load reduction before and after simulated aging. These tests provided objective data on the material’s actual ability to maintain its antimicrobial effect after exposure to demanding conditions.
E. coli was used as the model microorganism due to its widespread use in antimicrobial evaluation standards. The samples were tested following the Japanese Industrial Standard JIS Z 2801 method, as well as ISO 22196:2011 and ISO 20743:2013.
Quantitative microbiological validation allows differentiation between temporary efficacy loss and actual material degradation.
Variations in microbiological reduction were analyzed, and acceptance thresholds were established in alignment with the client’s technical requirements. This evaluation made it possible to distinguish between minor efficacy fluctuations and significant performance degradation.
INFINITIA’s Forensic Engineering team coordinated experimental execution, variable control, and critical interpretation of the results. The integration of quantitative data with the application context enabled well-founded and technically justified conclusions.
Benefits of technically validated material homologation
As a result, the client obtained a solid technical basis for deciding on the material’s suitability for its final application. The combination of accelerated testing, use simulation, and microbiological verification reduced uncertainty and anticipated potential performance deviations before large-scale production.
Material homologation was supported by reproducible experimental evidence, facilitating its defense in internal audits, regulatory requirements, or certification processes. Additionally, the study enabled the identification of possible formulation adjustments or design improvements prior to final implementation.
This approach not only validates a specific property but also strengthens overall product reliability in highly demanding environments, minimizing technical and economic risks associated with decisions based solely on preliminary data.