What was the challenge or problem to be solved?
In the electronics industry, the reliability of components largely depends on the quality of the materials and the coatings used to protect them from aggressive environments. Factors such as humidity, salinity, or the presence of contaminants can accelerate degradation processes, affecting the integrity of metallic connections and compromising the performance of devices over time.
In this context, an industrial company needed to validate a new supplier of components used in electronic boards. To make a technically sound decision, it was necessary to objectively evaluate how the coatings performed under demanding corrosive conditions.
Evaluation of corrosion in electronic components
Electronic components used in industrial environments can be exposed to demanding environmental conditions, where the presence of moisture, salts, or contaminants promotes corrosion processes. When these phenomena affect critical metallic elements, they can lead to connection failures, loss of conductivity, or progressive degradation of functional surfaces.
In this specific case, the client needed to evaluate the behavior of certain electronic components intended for integration into circuit boards. The main concern was the possible development of corrosion phenomena that could compromise the durability of the system under real operating conditions.
To address this issue, a comparative evaluation between components from two different suppliers was required. The objective was to determine whether significant differences existed in their behavior in corrosive environments and whether these differences could affect the reliability of the final product.
Evaluation of corrosion resistance
Corrosion resistance of metallic components is a critical factor in the design and manufacturing of electronic equipment intended for demanding environments. Surface coatings applied to metals act as protective barriers against aggressive agents, reducing the likelihood of degradation throughout the product’s service life.
However, small variations in manufacturing processes or coating quality can lead to significant differences in corrosion performance. These variations are not always visible to the naked eye and may only become apparent when the component is subjected to severe environmental conditions.
Corrosion resistance is not always visible: it must be validated through testing.
For this reason, the client needed reliable technical data to assess the level of protection provided by each supplier. A rigorous corrosion resistance analysis would help identify potential weaknesses in the coatings and support the supplier selection process with objective information.
Analysis of metallic coatings in electronic components
The study of metallic coatings is a key tool for understanding how components behave when exposed to degradation processes. These coatings may be composed of different materials or present varying thicknesses, factors that directly influence their ability to protect against corrosive environments.
In the analyzed case, the challenge consisted of determining whether structural or morphological differences existed between the coatings applied by each supplier. This required a detailed analysis of the most critical metallic areas of the component and an evaluation of their response after exposure to a controlled corrosive environment.
Through its Forensic Engineering area, INFINITIA proposed a comparative study aimed at identifying possible signs of degradation in the coatings and evaluating their protective capacity. The challenge involved not only reproducing aggressive environmental conditions but also performing a detailed characterization of the metallic surfaces to detect differences between suppliers.

How was it addressed or what was the solution?
To carry out the study, a testing methodology was designed to reproduce corrosive conditions in a controlled manner and evaluate the behavior of the electronic components with a high level of detail. The objective was to expose the samples to an aggressive environment that would accelerate degradation processes and allow differences between the analyzed metallic coatings to be identified.
The strategy combined standardized testing techniques with advanced microscopic characterization methods, enabling the analysis of both the surface evolution of corrosion and the internal structure of the coatings after exposure to severe conditions.
Accelerated corrosion testing to evaluate component behavior
The overall approach of the study was based on the application of an accelerated corrosion test, a methodology widely used in the industrial field to evaluate the durability of materials and coatings within a reduced period of time. This type of testing allows the simulation of aggressive environmental conditions that could occur during the product’s service life.
Before starting the test, the samples underwent an initial preparation stage to remove potential surface contaminants that could interfere with the analysis, ensuring that the surface of the components was in suitable condition for exposure to the corrosive environment.
This approach ensured that any differences observed during the test were related to the actual properties of the coatings rather than external factors. As a result, a reliable basis was established for comparing components from the different suppliers.
Salt spray testing as a comparative evaluation method
The central phase of the study involved performing a salt spray test, a standardized technique used to evaluate the corrosion resistance of metallic materials and coatings by exposing them to a highly corrosive environment.
During this test, the samples are placed in a chamber where a salt-rich controlled atmosphere is generated, reproducing conditions similar to those found in industrial or marine environments. This procedure accelerates degradation processes and makes it possible to observe, within a relatively short period of time, how materials respond to corrosion.
Salt spray testing enables comparison of coatings under controlled extreme conditions.
In this project, the salt spray test subjected the electronic components to severe exposure conditions, creating a suitable scenario to evaluate the behavior of the metallic coatings. Thanks to this comparative approach, differences in the corrosion evolution among the analyzed samples could be identified.
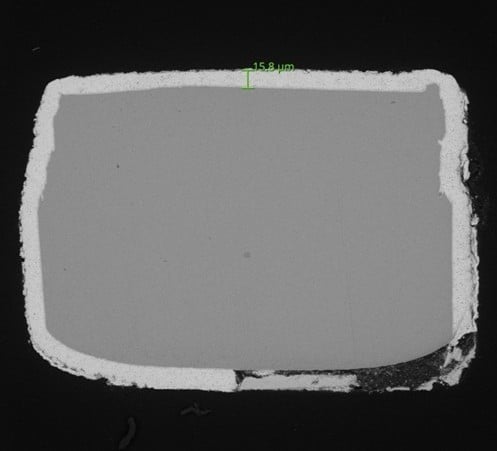
Analysis using scanning electron microscopy (SEM)
After completing the corrosion test, the samples underwent a preparation phase that included cleaning and metallographic processes. This stage removed salt residues generated during the test and prepared cross-sections of the components to facilitate microscopic observation.
The analysis was then carried out using scanning electron microscopy (SEM), a characterization technique that allows the morphology of surfaces to be observed with a high level of detail. This tool is particularly useful for identifying degradation phenomena, coating defects, or areas where corrosion has begun to develop.
SEM analysis reveals defects that are not visible to the naked eye.
Thanks to the use of scanning electron microscopy (SEM), the INFINITIA team was able to closely examine the critical metallic areas of the components and evaluate the impact of the corrosive environment on the coatings. This analysis made it possible to identify relevant differences in the behavior of samples from the different suppliers.
Based on this detailed characterization, a technical report was prepared summarizing the main observations of the study and comparing the performance of the analyzed coatings. As a result, the client was able to determine which supplier offered higher corrosion resistance, enabling a technically sound decision based on objective data and reducing the risk of premature failures in their electronic systems.